Clinical Trial Study Start-Up
Accelerating Clinical Trial Initiations with Customized Study Start-Up Solutions
Our clinical research team manages study start-up, site selection, and trial conduct across North America and with partner sites worldwide. We coordinate resources to keep projects moving safely and efficiently.
We handle regulatory submissions, engage with ethics committees, and manage site contracts and budgets. With experience across diverse sponsors and studies, we manage the start-up process effectively and support trials from planning through execution.
Clinical Trial Study Start-Up Strategy for Successful Initiation
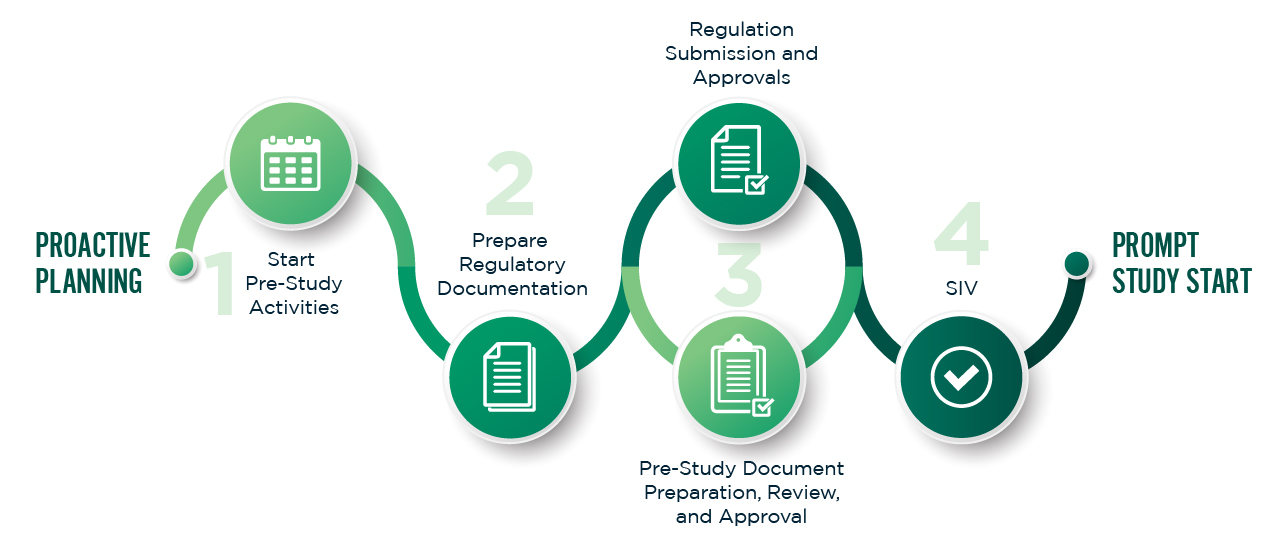
Our project team manages study start-up efficiently, defining planning elements early, completing reviews quickly, and securing required approvals from regulatory bodies. This ensures clinical activities begin on schedule.
Our start-up services include:
- developing project plans and timelines
- preparing essential documents and contracts for site initiation
- addressing regulatory requirements and potential hurdles
- coordinating with sites across North America and partner locations
- maintaining clear communication through centralized program management
- managing budgets, contracts, and vendor selection
Managing Site Activations
Because familiarity and continuity improve study start-up, we assign a designated project manager and a dedicated team of specialists for the full lifecycle of your trial. Together, they set timelines for each site and keep the project on track. This approach ensures consistency, even when amendments or new sites are added.
Explore our process for identifying, selecting, and managing clinical trials on the following pages: